|
3/17/2024 0 Comments Bohr model of atoms 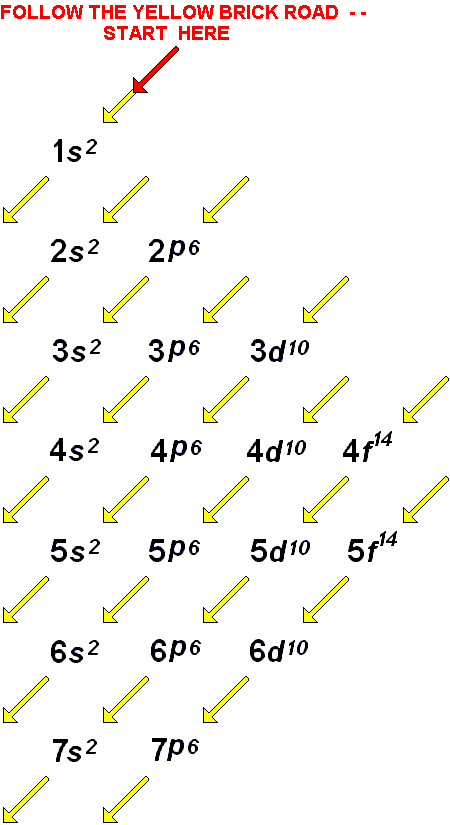
He carried out experiments using cathode rays produced in a discharge tube and found that the rays were attracted by positively charged metal plates but repelled by negatively charged ones. The first breakthrough came in the late 1800s when English physicist Joseph John (JJ) Thomson discovered that the atom wasn’t as indivisible as previously claimed. What followed was another, shorter lull where our knowledge of atoms didn’t progress all that much. There were some attempts to define what atoms might look like, such as Lord Kelvin’s suggestion that they might have a vortex-like structure, but it wasn’t until just after the turn of the 20th Century that progress on elucidating atomic structure really started to pick up. He came up with theories about how atoms combine to make compounds and also came up with the first set of chemical symbols for the known elements.ĭalton’s outlining of atomic theory was a start, but it still didn’t really tell us much about the nature of atoms themselves. However, since the neutron wouldn’t be discovered until 1932, we can probably forgive Dalton for this oversight. The latter point is one that pretty much still holds true, with the notable exception being isotopes of different elements, which differ in their number of neutrons. He drew on the ideas of the Ancient Greeks in describing atoms as small, hard spheres that are indivisible, and that atoms of a given element are identical to each other. It wasn’t until 1803 that the English chemist John Dalton started to develop a more scientific definition of the atom. It was a long wait, however, before these foundations were built upon. Though we now know that this is not the case, their ideas laid the foundations for future atomic models. Water atoms were smooth and slippery, explaining why water was a liquid at room temperature and could be poured. They envisaged iron atoms as having hooks which locked them together, explaining why iron was a solid at room temperature. These scholars imagined atoms as varying in shape depending on the type of atom. Though their ideas about atoms were rudimentary compared to our concepts today, they outlined the idea that everything is made of atoms, invisible and indivisible spheres of matter of infinite type and number. The Ancient Greek theory has been credited to several different scholars but is most often attributed to Democritus (460–370 BC) and his mentor Leucippus. The word ‘atom’ actually comes from Ancient Greek and roughly translates as ‘indivisible’. In fact, we have to go all the way back to Ancient Greece to find its genesis. Though our graphic starts in the 1800s, the idea of atoms was around long before. 
This graphic takes a look at the key models proposed for the atom, and how they changed over time. Despite this, our ideas about what an atom is are surprisingly recent: as little as one hundred years ago, scientists were still debating what exactly an atom looked like. This is something we now take as a given and one of the things you learn right back at the beginning of high school or secondary school chemistry classes. In fact Nield found that the ratio of energy in electrons and the frequency of their orbits around the nucleus was equal to Planck's constant (the proportion of light's energy to its wave frequency, or approximately 6.626 x 10-23 ).All matter is made up of atoms. This approach allowed Niels to explain a relatively stable atomic Model, which still hinged on the orbital model that Rutherford postulated. 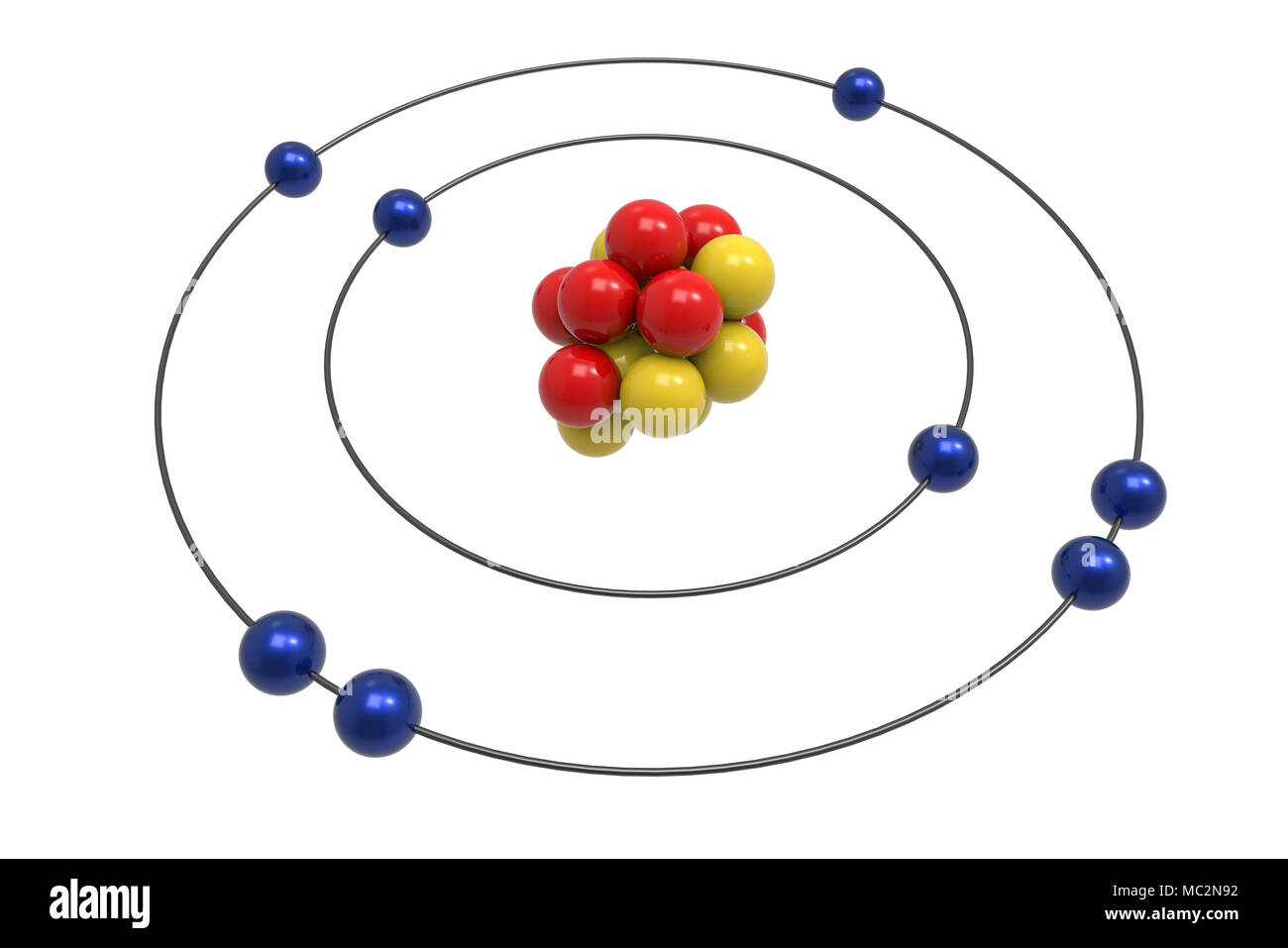
Niels Bohr which after his PhD circa 1912 had joined Rutherford realized that Rutherford's model wasn't quite right and started to articulate a model that borrowed from Planck's quantum theory. All of the negative charge was held in the electrons, orbiting the dense nucleus like planets around the sun. 
This model depicted an atomic model with nearly all its mass, and positive charge, in a central nucleus about 10,000 times smaller than the atom itself. Rutherford experiment with alpha particles shot at a thin gold foil resulted in the Rutherford model of the atom (Orbital Model). Thomson actually) which blew a big hole in "Plum Pudding Hypothesis" of the atom. Thomson that resulted "Plum Pudding" model of the atom and the 2nd one by Rutherford (a student of J.J. Well there were two experiments back to back one by J.J. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
